Product Sustenance Engineering
Overview
- Refers to ongoing support, maintenance, and improvement of software, hardware, and applications.
- Ensures products continue to operate effectively, reliably, and securely over time.
- Focuses on regulatory compliance, performance, and user needs throughout the product lifecycle.
Silmates Sustenance Engineering Expertise
- Specialized in Product Sustenance Engineering for medical devices.
- Services include:
- Design optimization for better efficiency.
- Regulatory compliance updates (FDA, ISO, MDR, etc.).
- Component obsolescence management to extend product life.
- Performance enhancement for reliability and safety.
- Ensures medical devices remain reliable, compliant, safe, and competitive in a dynamic healthcare industry.

Process Flow Diagram
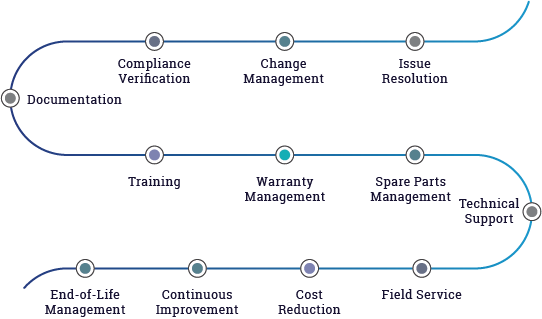
Key Offerings
Product Maintenance and Upgrades
Life Cycle Extension
Cost Reduction and Cost Optimization
Design Changes and Enhancements
Problem Resolution and Root Cause Analysis
Regulatory Compliance and Certification Support
